The density of 2.45 M aqueous methanol `(CH_(3)OH)` is `0.976 g/mL`. What is the molatiy of the sol - YouTube

OneClass: A solution is prepared by mixing 631 mL of methanol with 501 mL of water. The molarity of m...

Calculate the amount of benzoic acid (C6H5COOH) required for preparing 250 mL of 0.15 M solution in methanol.
Processes | Free Full-Text | Methanol in Grape Derived, Fruit and Honey Spirits: A Critical Review on Source, Quality Control, and Legal Limits

At 293.2K, the density of a 60% aqueous solution of methanol is 0.8946 g/mL. Calculate volume of 1 mole of the solution.

SOLVED: A solution is prepared by dissolving 20.2 mL of methanol (CH3OH) in 100.0 mL of water at 25 C The final volume of the solution is 118 mL. The densities of

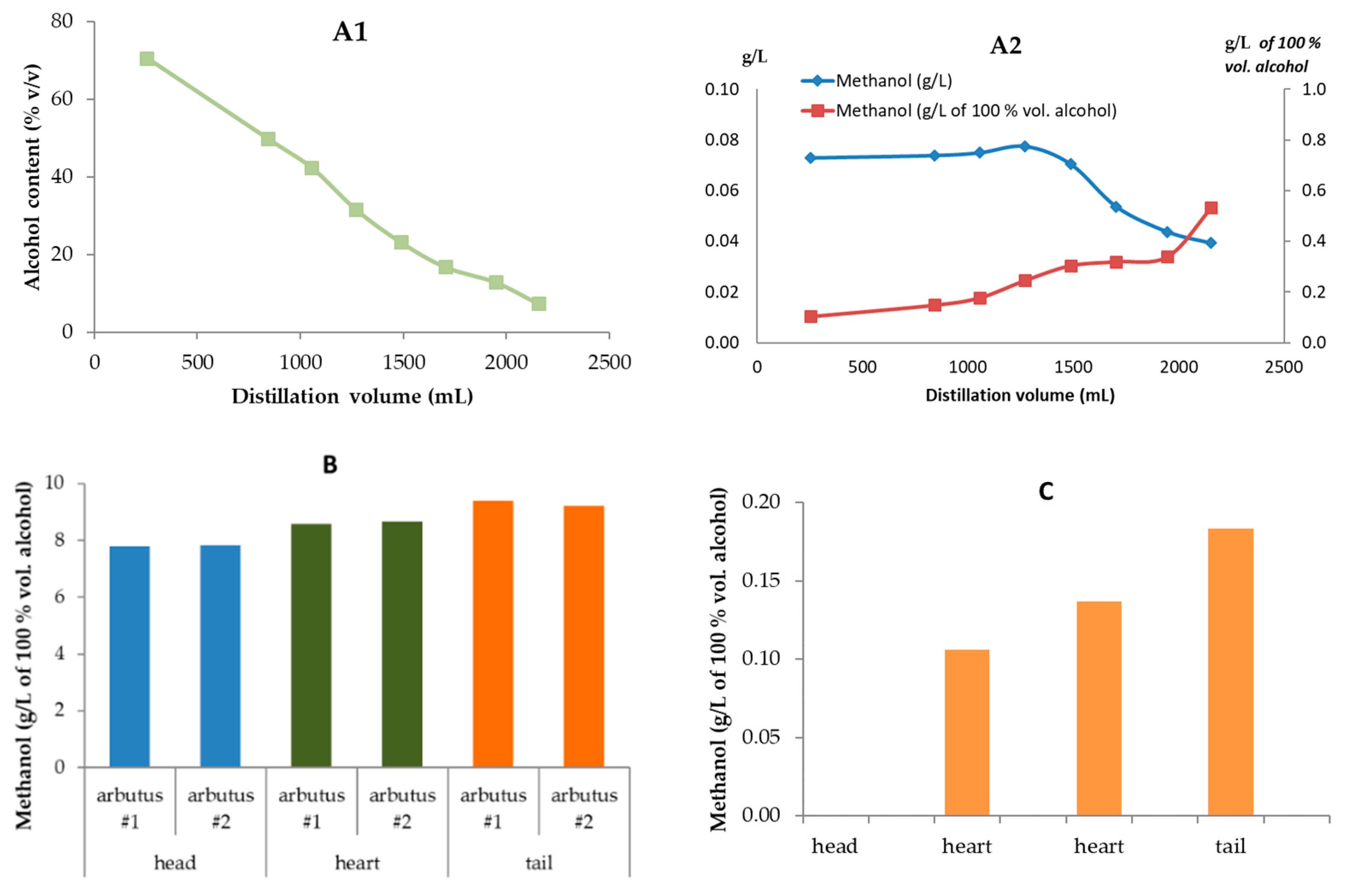
Methanol content (in grams per liter of 100% vol. alcohol, % and g per... | Download Scientific Diagram

SOLVED:The density of methanol, a colorless organic liquid used as solvent, is 0.7918 g / mL. Calculate the mass of 89.9 mL of the liquid.

Results of TPC calculation of antibacterial activity of gonad methanol... | Download Scientific Diagram

Exercise1.pdf - Methanol has a density of 2.79 g/mL. If 16.5 mL of this methanol is added to water to make a 2000.0 mL solution, calculate the | Course Hero

Calculate the molarity (M) and molality (m) of 16% aqueous methanol (CH(3) OH) solution by volume. Density of solution = 0.9 g mL^(-1).
The solubility of urea in methanol is 17 g urea/100 mL methanol. Density ofmethanol is 0.792 g/mL at 293 K at which vapour pressure of methanol is96.0 mm of Hg. Thus, vapour






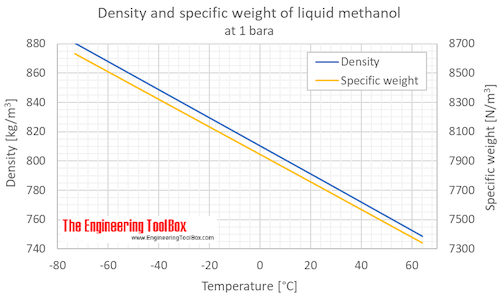
![ANSWERED] Calculate the osmotic pressure of each of... - Physical Chemistry ANSWERED] Calculate the osmotic pressure of each of... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/66524620-1657314910.1337452.jpeg)